  
This is because a new electron shell is added per each period when we go downward a group. As we move down a group in the periodic table, the ionic radius is increased. Same as in atomic radius, the ionic radius also has trends in the periodic table. The most common method to find the ionic radius is the X-ray crystallography. The ions of the same chemical element can be found in different sizes according to their electrical charges. For example, if an ionic compound is composed of a cation and an anion that has an atomic size which is three times larger, the distance between the two nuclei should be divided by 4 in order to obtain the cation radius.įigure 2: Atomic and Ionic Radii of Some Elements 
The most accurate way to find the ionic radius is to divide the distance between two nuclei of two ions according to their sizes. The anion is large because it has extra electrons that are repelled by the nucleus, resulting in the increase of the distance between the nucleus and the farthest electron of the electron cloud. 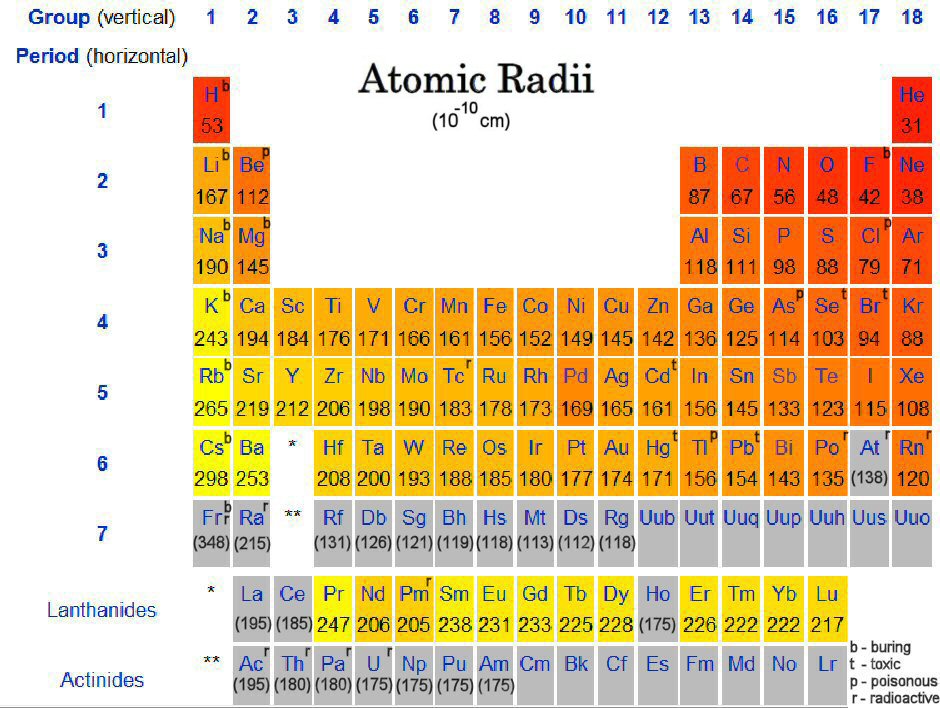
The cation is smaller in size because a cation is formed by removing one or more electrons from an atom. An ionic compound is composed of cations and anions. This compound is called an ionic compound because it is made out of ionic components. If it is a positively charged ion, it will react with a negatively charged ion (or the opposite) and become a stable neutral compound. Ionic radius is the radius of an atom’s ion. Likewise, when moving along a period, the size of the atom is decreased gradually, so is the atomic radius. It causes the size of the atom to shrink then the atomic radius is reduced. Therefore, the attraction force between electrons and the nucleus is high. But when moving along a period, the number of protons in the nucleus is increased along with the number of electrons present in the atom. Therefore, the atom is large, and the atomic radius is also large. Then the attraction from the nucleus is less. In the beginning of the period, there is a less number of electrons present in the outermost orbital. If the number of electrons present is higher, the attraction between the electrons and the nucleus is also high. The elements in the same period have the same number of electron shells. Along a period of the periodic table, the atomic number is gradually decreased. When considering the periodic table of elements, there is a pattern of the atomic radius of elements. Atomic radius can be defined only for isolated and neutral atoms. In other words, it is the distance from the nucleus to the farthest electron that belongs to that atom. Key Terms: Atomic Radius, Atoms, Electron Shell, Ionic Radius, Ions What is Atomic RadiusĪtomic radius is the distance from the nucleus of an atom to its boundary of the electron cloud. What is the Difference Between Atomic Radius and Ionic Radius – Definition, Trends in the Periodic Tableģ. The main difference between atomic radius and ionic radius is that atomic radius is the radius of a neutral atom whereas ionic radius is the radius of an electrically charged atom. The radius of an ion can be either larger or smaller than the radius of an atom, depending on the electrical charge of the ion. Atomic radius is the distance between the nucleus of an atom and the boundary of its electron cloud. Because an atom or ion is composed of electrons that are in movement. Since atoms and ions are circular 3D structures, we can measure the radius of an atom or an ion. These atoms can be converted into ions by adding one or more electrons from outside. Main Difference – Atomic Radius vs Ionic RadiusĪtoms are the building blocks of matter. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
